The MTHFR gene sits in a controversial space in both nutrition and medicine. On one end, it’s often linked to a wide range of health issues — from mental health and fatigue to fertility and cardiovascular risk. On the other, medical guidelines consistently recommend against routine testing, stating it rarely changes treatment.
In this post, we’ll unpack what MTHFR gene variants like C677T and A1298C mean, what the science says about their impact, and whether it’s something that’s truly worth testing or acting on.
What Is It?
MTHFR is an abbreviation that is used interchangeably for both a gene and an enzyme.
It stands for Methylenetetrahydrofolate reductase. You can see why everyone just uses the abbreviation.
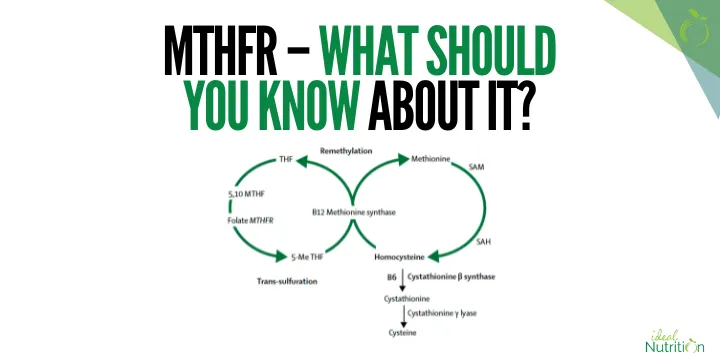
The MTHFR enzyme is involved in folate metabolism.
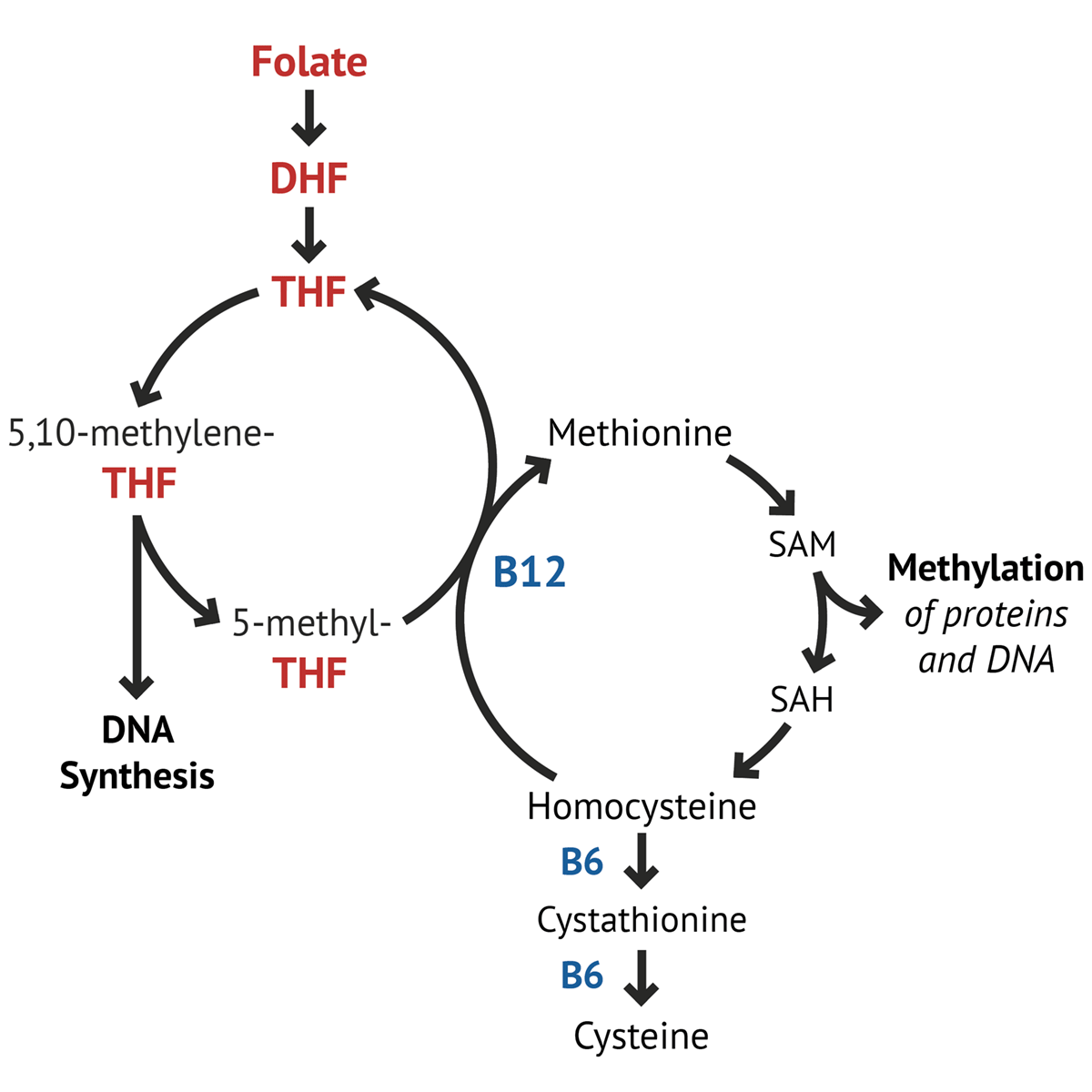
It helps turn folate into its active form, supports homocysteine breakdown, and fuels methylation — which underpins everything from cardiovascular health to gene expression.
The MTHFR gene provides the instructions to make the MTHFR enzyme.
There are over 40 known mutations (polymorphisms) in the MTHFR gene, but the main two commonly studied and discussed are:
- C677T
- A1298C
How Prevalent Are These Genetic Variations?
This was the first question that popped into my mind when I first heard about this topic many years ago. This will become more relevant when we look deeper as to how much these variations matter.
Roughly 40–60% of people globally have some form of these two variants. Often, I back up almost all my claims with research links, but this one is just a general estimate based on a combination of sources.
At face value, this would mean that if this topic mattered, it would matter for a lot of people.
Going one step deeper, there are different forms of variants.
Everyone has two copies of the MTHFR gene — one from each parent. When we talk about MTHFR “mutations” or “variants,” we’re talking about whether someone has zero, one, or two copies of a particular variant.
Having two copies of the variant can have a bigger impact on how well the MTHFR enzyme works.
Based on similar estimates:
- 30-50% of people have one copy of the C677T variant
- 10-15% have two copies
The prevalence is still quite high, even with two copies.
What Can Happen with These Genetic Variances and Why Might It Be Relevant?
When somebody has a variant, it can change how effectively the MTHFR enzyme works.
Since it plays a role in processing folate and regulating homocysteine levels, a lot of the relevance is related to those. Since this is a broad section, we will look at each area individually.
Elevated Homocysteine

MTHFR variants can contribute to elevated homocysteine, but they won’t always.
The MTHFR enzyme helps convert folate into its active form, which is needed to turn homocysteine into methionine.
If the enzyme activity is reduced, this process can slow down, leading to a buildup of homocysteine.
Elevated homocysteine is associated with things like:
– Heart attack, stroke, and atherosclerosis.
– Pregnancy complications
– Cognitive decline
Another stat that’s hard to pin down is that, reportedly, having two copies of the C677T leads to an average increase of 15–25% in plasma homocysteine levels.
Other factors, such as low intake of folate, B12, and B6, increase the risk of elevated homocysteine in people with and without these variations.
Pregnancy

In Australia, like many countries around the world, folic acid supplementation is recommended during pregnancy.
There is concern that because of the reduction in folate metabolism, folic acid supplementation (especially because it is synthetic rather than natural) will cause issues.
This concern mostly comes from the idea that people with MTHFR variants can’t properly convert folic acid into its active form, leading to a potential buildup of unmetabolised folic acid and downstream effects like impaired methylation or increased health risks. However, this is largely theoretical.
Arguably, this is the biggest concern when factoring in how prevalent these variations are.
In practice, even people with MTHFR variants can still convert folic acid into its active form, especially when the dosage is appropriate.
Folic acid has been found to reduce the risk of neural tube defects in large population studies and clinical trials.
Alternatives exist, such as methylfolate, which theoretically gets around this issue, particularly if homocysteine is elevated. But in a later section, we’ll cover the pros and cons of this. And we will also discuss what the recommendation is for people with the MTHFR gene variants in general.
Cardiovascular Disease
Since homocysteine has a link with cardiovascular disease, it makes sense that this is a risk.
There is some research linking MTHFR gene variants with outcomes like stroke and heart attacks.
Based on the mechanisms, the main risk is associated with elevated homocysteine.
Maintaining a good intake of folate, B6, and B12 (through either food or supplements) can help reduce this.
To the best of my knowledge, it hasn’t technically been studied whether increasing those things reduces the risk of cardiovascular events in those with MTHFR variants. It makes sense to do regardless, though.
Other Proposed Links
There are a lot of other proposed links with MTHFR gene variants and health outcomes. It can turn into an exhaustive list. Some of the concerns are relevant, and some are not. To keep this simple, I have just covered the above aspects, but there are still other relevant topics out there.
Proposed Solutions
The most common proposed solutions for those with MTHFR variants are:
- Switching to active forms of B Vitamins when supplementing
– Folic acid (folate) → methylfolate
– Cyanocobalamin (B12) → methylcobalamin
– Pyridoxine (B6) → pyridoxal-5-phosphate - Supplementing with B vitamins, even with normal diets.
- Test homocysteine levels.
- Adjust lifestyle:
– Reduce alcohol since it impairs folate absorption.
– Eat foods rich in folate.
– Manage stress, since it can deplete methylation co-factors.
Note that these are just proposed solutions and not necessarily recommendations from me. I just feel they are relevant to include before proceeding to the next section.
Why Is It Not Standard Medical Practice to Test for MTHFR Variations?
The gold standard guidelines for GPs typically recommend not testing for MTHFR variations outside of rare cases.

Directly quoting the Australian ones:
- “Avoid requesting an MTHFR gene test as there is no causal link between MTHFR gene variations and particular diseases, and no substantial evidence to support the use of MTHFR gene testing in routine clinical practice.
- There is no evidence-based treatment that will improve health outcomes for a patient who has one or both MTHFR gene variants, so knowledge of a patient’s MTHFR gene status is unlikely to change how you manage and treat the patient.
- Women of childbearing age should take the standard dose of folic acid supplementation to prevent neural tube defects during pregnancy, regardless of their MTHFR status.
- For patients who are carriers of the variants of the MTHFR gene, continue to provide normal care.”
If you had a strong belief or personal experience around MTHFR variations, this direct quote could seem negligent.
Response to Criticism of That Position
If I were to write my own response to criticism of that direct quote, as if I were writing on their behalf I would say:
- MTHFR variants (like C677T and A1298C) do affect folate metabolism to some degree, but the clinical significance is often overstated, especially in wellness/functional medicine circles.
- Large population studies haven’t shown a clear benefit of changing folate recommendations based on MTHFR status.
- Standard folic acid supplementation (500 μg/day) is still effective at preventing neural tube defects, even in those with common MTHFR polymorphisms.
- There’s no strong evidence that testing MTHFR status improves outcomes or warrants changes like switching to methylfolate for most people.
- If concerned about homocysteine levels and B vitamins, you can test those directly.
- Over-interpreting MTHFR status can lead to unnecessary anxiety or costly supplement use with no proven benefit.
Standard folic acid works and in the vast majority of cases, testing doesn’t change management.
Is There a Downside to Switching from Folic Acid to Methylfolate if You Prefer?
I think this is a very valid question and something that will be on the minds of a lot of women looking at pregnancy-related aspects.
The first part is that theoretically, there is no downside outside of cost. If cost isn’t a barrier here, that works well. The recommended dosage is the same.
If looking for downsides, the two main relevant ones are:
- There is far less research. We have extensive research showing that folic acid supplementation reduces neural tube defects in pregnancies with and without MTHFR variants. We don’t have nearly the same amount for methylfolate.
- It’s arguably higher risk if you take too high of a dosage. But this can be avoided just by sticking to the recommended dosage of ~400-500 mcg per day.
My best interpretation is that it is likely fine to make that switch if you want. It’s just that it doesn’t have the same evidence base due to a lack of research.
Rarer Exceptions
Some MTHFR variations exist that can cause very low to absent enzyme activity. Often (ideally) this is diagnosed in infancy or early childhood.
In those cases, the approach is very different. There is often far more aggressive nutrient support of things like betaine (to help clear homocysteine), methylfolate, methylcobalamin and a few other things, in addition to more frequent monitoring.
What Should You Do With This Information?
For most people, there’s no need to act on MTHFR status—especially if you feel well, have normal homocysteine levels, and get enough folate, B12, and B6.
That’s why routine testing isn’t recommended—it rarely changes treatment. That said, testing out of interest isn’t harmful.
If you do have a variant and any of the following apply:
- Elevated homocysteine
- Planning a pregnancy and want to be cautious
- Family or personal history of NTDs, CVD, or infertility
- Prefer using active B vitamins and can afford it
Then supplementing with active forms like methylfolate, methylcobalamin, and P-5-P is a reasonable option. It’s unlikely to cause harm at standard doses and may offer peace of mind.
A cautious medical perspective would still highlight that folic acid has more research behind it for preventing neural tube defects, especially since most of that data includes a large percentage of people with MTHFR variants.
If you haven’t tested and have no issues, there’s no strong reason to start now.
In short:
- Folic acid supplementation is proven and effective in the majority of cases still.
- Use methylfolate if you prefer—it’s a valid alternative.
- If you’re concerned, you can test homocysteine and B vitamin levels directly